Topic 1: Systems and models
Introduction
It is essential that the systems approach is used throughout this course. This approach identifies the elements of the systems, and examines the relationships and processes that link these elements into a functioning entity. This topic may be best viewed therefore as a theme to be used in the delivery of the other topics, rather than as an isolated teaching topic.
The topic identifies some of the underlying principles that can be applied to living systems, from the level of the individual up to that of the whole biosphere. It would therefore be helpful to describe and analyse the systems addressed in the terms laid out in this topic (wherever possible). The systems approach also emphasizes the similarities between environmental systems, biological systems and artificial entities such as transport and communication systems. This approach stresses that there are concepts, techniques and terms that can be transferred from one discipline (such as ecology) to another (such as engineering).
The topic identifies some of the underlying principles that can be applied to living systems, from the level of the individual up to that of the whole biosphere. It would therefore be helpful to describe and analyse the systems addressed in the terms laid out in this topic (wherever possible). The systems approach also emphasizes the similarities between environmental systems, biological systems and artificial entities such as transport and communication systems. This approach stresses that there are concepts, techniques and terms that can be transferred from one discipline (such as ecology) to another (such as engineering).
1.1.1 - Outline the concept and characteristics of systems.
A System is an assemblage of parts, working together, forming a functioning whole. There are environmental systems that we will discuss in this subject. Systems can also be political, economic, social etc When we talk about the environment, systems can be non living or living and might be very large or very small.
Biosphere = atmosphere + lithosphere + hydrosphere + ecosphere
|
The Earth: This system is made up of different parts interacting;
Atmosphere: The collection of gases above the Earths surface. Hydrosphere: The water on Earth in all its states; solid, liquid and gas. Lithosphere (Geosphere): Solid rock on the surface, molten rock below. Ecosphere (biosphere): All living things on Earth. |
Non-living system example: The Bike
Living system example: The Cell
|
1.1.2 - Apply the systems concept on a range of scales.
Ecosystems can be considered on a range of scales; small to large.
1.1.3 - Define the terms open system, closed system and isolated system.
Open Systems: exchange matter and energy with its surroundings. Most systems are open.
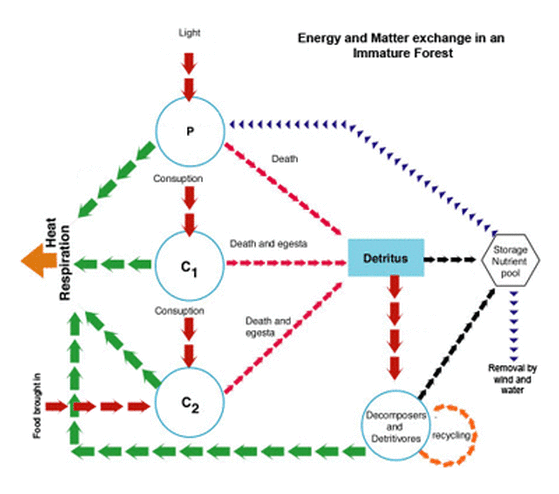
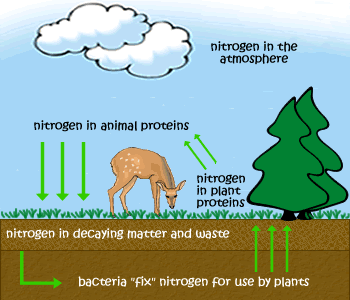
- Example: The Forest - Plants use light energy during photosynthesis, nitrogen from the air is fixed in the soil and used by plants for growth, herbivores eat the plants and then fertilise the soil with faeces, water is lost by evaporation and transpiration and replaced by rain.
Closed System: A system which exchanges energy but not matter with its environment. These types of systems are extrmemely rare on our planet.
- Example 1: The entire Earth - can be seen as a closed system (almost!). Light energy enters the Earth system. Some of this is released back into space as long-wave radiation.
- Example 2: Biosphere 2 - A man-made, closed city built in Arizona in the 1980s constructed to explore the use of closed biospheres in space colonization. It was not successful as it could not produce enough food and oxygen levels became too low.
Isolated System: Type of a system which doesn’t exchange matter or energy with surrounding environment. Some consider the entire universe as an isolated system.
1.1.4 - Describe how the first and second laws of thermodynamics are relevant to environmental systems.
|
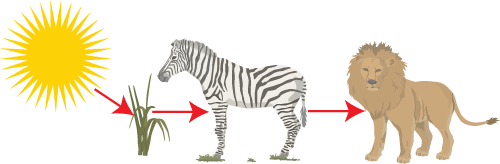
First Law of Thermodynamics
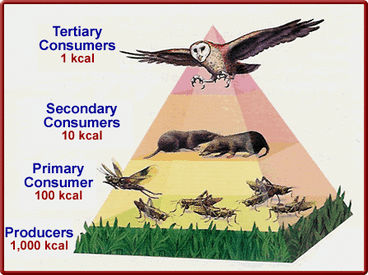
Energy is neither created or destroyed. First law of thermodynamics is same as conservation of energy In the picture opposite we see the simple food chain in which energy is transferred from sunlight to the plants later to Zebra and then to the carnivore. The solar energy gets converted to stored chemical and later the chemical energy (glucose) is passed along as food so new energy is not created or destroyed. |
Second Law of Thermodynamics
The entropy of an isolated system not in equilibrium will tend to increase over time. Energy conversions are never 100%, some energy is always lost to the envionment as heat. Energy = work + heat (and other wasted energy)
|
1.1.5 - Explain the nature of equilibria.
|
Equilibrium: the tendency of a system (particularly open systems) to return to an original state following disturbance; at equilibrium , a state of balance exists among all of the components of that system. Systems do change, but these changes tend to be within limits.
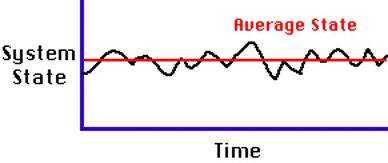
Steady-state equilibrium: When there is a continuous input and output of energy and matter, but the system remains in a relatively constant state. There are short term fluctuations, but no long-term changes.
Static-state equilibrium: When there is no change over time. components of the system do not change their position or state over long periods of time.
|
1.1.6 - Define and explain the principles of positive feedback and negative feedback.
|
Positive feedback: A feedback system that tends to amplify the output of the system.
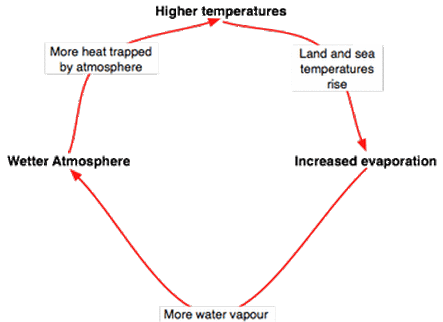
Positive feedback is unstable unlikely to Negative feedback, it has an exponential deviation which means only amplifying or increasing something and returning to the previous position. Example: Global warming- The diagram opposite shows that global warming is an example of positive feedback. More water vapor leads to a wetter atmosphere and more heat trapped by the atmosphere. More trapped heat increases the temperature of the land, sea and atmosphere and increased evaporation |
|
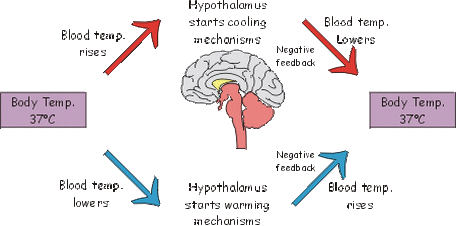
Negative Feedback: A feedback in which the system responds in an opposite direction to the original change.
Example: Your body heats up on a hot day. The sensors in your skin detect the rising temperature. As a result you start to sweat and blood flows to the skin capillaries to lose heat. Your body loses heat. |
1.1.7 - Describe transfer and transformation processes.
Both matter (materials) and energy flow through an Ecosystem. This flow can be described as either a transfer or transformation. Both types require energy.
|
Transfer: when the flow does not involve a chance is state but simply the movement of material or energy from one place to another. Transfers require less energy (more efficient) than Transformations because they are simpler processes.
For example;
|
Transformation: when a flow involves a change in state.
For example;
|
1.1.8 - Distinguish between flows (inputs and outputs) and storages (stock) in relation to systems.
|
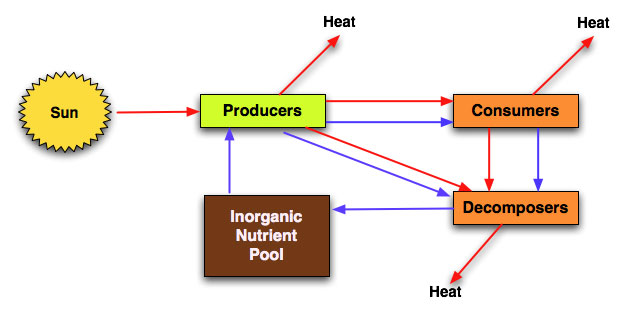
Flows: Energy and matter both flow through an ecosystem.
Stock: At different stages both energy and matter can be stored, the place where they are stored is called a stock. For example, there is a stock of chemical potential energy in the flesh of animals. This same flesh is composed primarily of proteins. The amino acids making up these proteins contains a nitrogen stock. In systems diagrams, flows are represented by arrows and stores by boxes
|
Ecosystems also contain;
|
1.1.9 - Construct and analyse quantitative models involving flows and storages in a system.
1.1.10 - Evaluate the strengths and limitations of models.
|
Strengths
|
Weaknesses
|